There is a feedback form at the bottom of this page. Please must fill it out.
इस पृष्ठ के नीचे एक फीडबैक फॉर्म है। कृपया इसे अवश्य भरें।
इस पृष्ठ के नीचे एक फीडबैक फॉर्म है। कृपया इसे अवश्य भरें।
communication English -1
UNIT- 1
UNIT - II
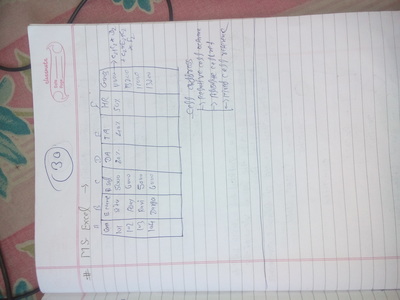
COMPUTER
|
|
|
| ||||||||||||||||||||||||||||||
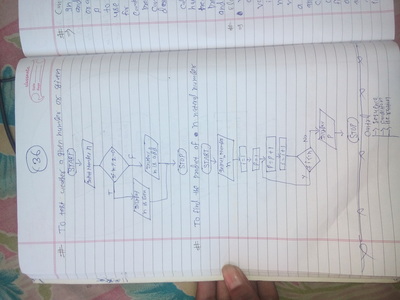
Engineering mathematics
Cramer's Rule
Given a system of linear equations, Cramer's Rule is a handy way to solve for just one of the variables without having to solve the whole system of equations. They don't usually teach Cramer's Rule this way, but this is supposed to be the point of the Rule: instead of solving the entire system of equations, you can use Cramer's to solve for just one single variable.
Let's use the following system of equations:
- 2x + y + z = 3
x – y – z = 0
x + 2y + z = 0
2x + 1y + 1z = 3
1x – 1y – 1z = 0
1x + 2y + 1z = 0
- x = 3/3 = 1, y = –6/3 = –2, and z = 9/3 = 3
- Given the following system of equations, find the value of z.
- 2x + y + z = 1
x – y + 4z = 0
x + 2y – 2z = 3
- z = 2
Almost.
What if the coefficient determinant is zero? You can't divide by zero, so what does this mean? I can't go into the technicalities here, but "D = 0" means that the system of equations has no unique solution. The system may be inconsistent (no solution at all) or dependent (an infinite solution, which may be expressed as a parametric solution such as "(a, a + 3, a – 4)"). In terms of Cramer's Rule, "D = 0" means that you'll have to use some other method (such as matrix row operations) to solve the system. If D = 0, you can't use Cramer's Rule.
matrices
Definition;-
A matrix is a rectangular array of numbers or other mathematical objects, for which operations such as addition and multiplication are defined. Most commonly, a matrix over a field F is a rectangular array of scalars from F. Most of this article focuses on real and complex matrices, i.e., matrices whose elements are real numbers or complex numbers, respectively. More general types of entries are discussed below. For instance, this is a real matrix:
types of matrices
A row matrix is a matrix with only one row.
A column matrix is a matrix with only one column.
A zero matrix or a null matrix is a matrix that has all its elements zero.
A square matrix is a matrix with an equal number of rows and columns.
A unit matrix is a diagonal matrix whose elements in the diagonal are all ones.
A matrix is a rectangular array of numbers or other mathematical objects, for which operations such as addition and multiplication are defined. Most commonly, a matrix over a field F is a rectangular array of scalars from F. Most of this article focuses on real and complex matrices, i.e., matrices whose elements are real numbers or complex numbers, respectively. More general types of entries are discussed below. For instance, this is a real matrix:
types of matrices
A row matrix is a matrix with only one row.
A column matrix is a matrix with only one column.
A zero matrix or a null matrix is a matrix that has all its elements zero.
A square matrix is a matrix with an equal number of rows and columns.
A unit matrix is a diagonal matrix whose elements in the diagonal are all ones.
Binomial Theorem
| binomial_theorem.pdf | |
| File Size: | 71 kb |
| File Type: | |
Complex Number
| complex_number.pdf | |
| File Size: | 178 kb |
| File Type: | |
| demoivers_theorem.pdf | |
| File Size: | 111 kb |
| File Type: | |
| analytical_geometry.pdf | |
| File Size: | 5530 kb |
| File Type: | |
| trigonometry_notes.pdf | |
| File Size: | 1566 kb |
| File Type: | |
| limits_and_continuti.pdf | |
| File Size: | 1211 kb |
| File Type: | |
Engineering Physics
| |||||||
| |||||||
| |||||||
| |||||||
| |||||||
| |||||||
Engineering Chemistry
Water chemistry
Organic chemistry
Fundamentals of thermodynamics
Definition
Thermodynamics is the branch of physics that deals with the relationships between heat and other forms of energy. In particular, it describes how thermal energy is converted to and from other forms of energy and how it affects matter.
Law's of thermodynamics
The four laws of thermodynamics The fundamental principles of thermodynamics were originally expressed in three laws. Later, it was determined that a more fundamental law had been neglected, apparently because it had seemed so obvious that it did not need to be stated explicitly. To form a complete set of rules, scientists decided this most fundamental law needed to be included. The problem, though, was that the first three laws had already been established and were well known by their assigned numbers. When faced with the prospect of renumbering the existing laws, which would cause considerable confusion, or placing the per-eminent law at the end of the list, which would make no logical sense, a British physicist, Ralph H. Fowler, came up with an alternative that solved the dilemma: he called the new law the “Zeroth Law.” In brief, these laws are:
The Zeroth Law states that if two bodies are in thermal equilibrium with some third body, then they are also in equilibrium with each other. This establishes temperature as a fundamental and measurable property of matter.
The First Law states that the total increase in the energy of a system is equal to the increase in thermal energy plus the work done on the system. This states that heat is a form of energy and is therefore subject to the principle of conservation.
The Second Law states that heat energy cannot be transferred from a body at a lower temperature to a body at a higher temperature without the addition of energy. This is why it costs money to run an air conditioner.
The Third Law states that the entropy of a pure crystal at absolute zero is zero. As explained above, entropy is sometimes called "waste energy," i.e., energy that is unable to do work, and since there is no heat energy whatsoever at absolute zero, there can be no waste energy. Entropy is also a measure of the disorder in a system, and while a perfect crystal is by definition perfectly ordered, any positive value of temperature means there is motion within the crystal, which causes disorder. For these reasons, there can be no physical system with lower entropy, so entropy always has a positive value.
Exothermic & Endothermic reactions
An exothermic reaction is a chemical or physical reaction that releases heat. It gives net energy to its surroundings. That is, the energy needed to initiate the reaction is less than the energy that is subsequently released.
When the medium in which the reaction is taking place gains heat, the reaction is exothermic. When using a calorimeter, the total amount of heat that flows into (or through) the calorimeter is the negative of the net change in energy of the system.
Examples of exothermic reactions
Endothermic process
the term endothermic process describes a process or reaction in which the system absorbs energy from its surroundings; usually, but not always, in the form of heat. The term was coined by Marcellin Berthelot from the Greek roots endo-, derived from the word "endon" (ἔνδον) meaning "within" and the root "therm" (θερμ-) meaning "hot." The intended sense is that of a reaction that depends on absorbing heat if it is to proceed. The opposite of an endothermic process is an exothermic process, one that releases, "gives out" energy in the form of (usually, but not always) heat. Thus in each term (endothermic & exothermic) the prefix refers to where heat goes as the reaction occurs, though in reality it only refers to where the energy goes, without necessarily being in the form of heat.
The concept is frequently applied in physical sciences to, for example, chemical reactions, where thermal energy (heat) is converted to chemical bond energy.
Endothermic (and exothermic) analysis only accounts for the enthalpy change (∆H) of a reaction. The full energy analysis of a reaction is the Gibbs free energy (∆G), which includes an entropy (∆S) and temperature term in addition to the enthalpy. A reaction will be a spontaneous process at a certain temperature if the products have a lower Gibbs free energy (an exergonic reaction) even if the enthalpy of the products is higher. Entropy and enthalpy are different terms, so the change in entropic energy can overcome an opposite change in enthalpic energy and make an endothermic reaction favorable.
Examples
Expansion of 1st law of thermodynamics
The first law of thermodynamics is a version of the law of conservation of energy, adapted for thermodynamic systems. The law of conservation of energy states that the total energy of an isolated system is constant; energy can be transformed from one form to another, but cannot be created or destroyed. The first law is often formulated by stating that the change in the internal energy of a closed system is equal to the amount of heat supplied to the system, minus the amount of work done by the system on its surroundings. Equivalently, perpetual motion machines of the first kind are impossible.
Adiabatic processes
In an adiabatic process, there is transfer of energy as work but not as heat. For all adiabatic process that takes a system from a given initial state to a given final state, irrespective of how the work is done, the respective eventual total quantities of energy transferred as work are one and the same, determined just by the given initial and final states. The work done on the system is defined and measured by changes in mechanical or quasi-mechanical variables external to the system. Physically, adiabatic transfer of energy as work requires the existence of adiabatic enclosures
Thermodynamics is the branch of physics that deals with the relationships between heat and other forms of energy. In particular, it describes how thermal energy is converted to and from other forms of energy and how it affects matter.
Law's of thermodynamics
The four laws of thermodynamics The fundamental principles of thermodynamics were originally expressed in three laws. Later, it was determined that a more fundamental law had been neglected, apparently because it had seemed so obvious that it did not need to be stated explicitly. To form a complete set of rules, scientists decided this most fundamental law needed to be included. The problem, though, was that the first three laws had already been established and were well known by their assigned numbers. When faced with the prospect of renumbering the existing laws, which would cause considerable confusion, or placing the per-eminent law at the end of the list, which would make no logical sense, a British physicist, Ralph H. Fowler, came up with an alternative that solved the dilemma: he called the new law the “Zeroth Law.” In brief, these laws are:
The Zeroth Law states that if two bodies are in thermal equilibrium with some third body, then they are also in equilibrium with each other. This establishes temperature as a fundamental and measurable property of matter.
The First Law states that the total increase in the energy of a system is equal to the increase in thermal energy plus the work done on the system. This states that heat is a form of energy and is therefore subject to the principle of conservation.
The Second Law states that heat energy cannot be transferred from a body at a lower temperature to a body at a higher temperature without the addition of energy. This is why it costs money to run an air conditioner.
The Third Law states that the entropy of a pure crystal at absolute zero is zero. As explained above, entropy is sometimes called "waste energy," i.e., energy that is unable to do work, and since there is no heat energy whatsoever at absolute zero, there can be no waste energy. Entropy is also a measure of the disorder in a system, and while a perfect crystal is by definition perfectly ordered, any positive value of temperature means there is motion within the crystal, which causes disorder. For these reasons, there can be no physical system with lower entropy, so entropy always has a positive value.
Exothermic & Endothermic reactions
An exothermic reaction is a chemical or physical reaction that releases heat. It gives net energy to its surroundings. That is, the energy needed to initiate the reaction is less than the energy that is subsequently released.
When the medium in which the reaction is taking place gains heat, the reaction is exothermic. When using a calorimeter, the total amount of heat that flows into (or through) the calorimeter is the negative of the net change in energy of the system.
Examples of exothermic reactions
- Combustion reactions of fuels
- Neutralization
- Burning of a substance
- Adding water to anhydrous copper(II) sulfate
- The thermite reaction
- Reactions taking place in a self-heating can based on lime aluminium
- Many corrosion reactions such as oxidation of metals
- Most polymerization reactions
- The Haber process of ammonia production
- Respiration
- Decomposition of vegetable matter into compost
- Most of the acid based reactions are exothermic
Endothermic process
the term endothermic process describes a process or reaction in which the system absorbs energy from its surroundings; usually, but not always, in the form of heat. The term was coined by Marcellin Berthelot from the Greek roots endo-, derived from the word "endon" (ἔνδον) meaning "within" and the root "therm" (θερμ-) meaning "hot." The intended sense is that of a reaction that depends on absorbing heat if it is to proceed. The opposite of an endothermic process is an exothermic process, one that releases, "gives out" energy in the form of (usually, but not always) heat. Thus in each term (endothermic & exothermic) the prefix refers to where heat goes as the reaction occurs, though in reality it only refers to where the energy goes, without necessarily being in the form of heat.
The concept is frequently applied in physical sciences to, for example, chemical reactions, where thermal energy (heat) is converted to chemical bond energy.
Endothermic (and exothermic) analysis only accounts for the enthalpy change (∆H) of a reaction. The full energy analysis of a reaction is the Gibbs free energy (∆G), which includes an entropy (∆S) and temperature term in addition to the enthalpy. A reaction will be a spontaneous process at a certain temperature if the products have a lower Gibbs free energy (an exergonic reaction) even if the enthalpy of the products is higher. Entropy and enthalpy are different terms, so the change in entropic energy can overcome an opposite change in enthalpic energy and make an endothermic reaction favorable.
Examples
- Photosynthesis
- Melting ice
- Cracking of alkanes
- Vaporising rubbing alcohol
- Thermal decomposition reactions
- Dissolving ammonium chloride in water
Expansion of 1st law of thermodynamics
The first law of thermodynamics is a version of the law of conservation of energy, adapted for thermodynamic systems. The law of conservation of energy states that the total energy of an isolated system is constant; energy can be transformed from one form to another, but cannot be created or destroyed. The first law is often formulated by stating that the change in the internal energy of a closed system is equal to the amount of heat supplied to the system, minus the amount of work done by the system on its surroundings. Equivalently, perpetual motion machines of the first kind are impossible.
Adiabatic processes
In an adiabatic process, there is transfer of energy as work but not as heat. For all adiabatic process that takes a system from a given initial state to a given final state, irrespective of how the work is done, the respective eventual total quantities of energy transferred as work are one and the same, determined just by the given initial and final states. The work done on the system is defined and measured by changes in mechanical or quasi-mechanical variables external to the system. Physically, adiabatic transfer of energy as work requires the existence of adiabatic enclosures
Rubber
Fuels
Electro chemistry
Engineering Materials
Abrasive:-
An abrasive is a material, often a mineral, that is used to shape or finish a workpiece through rubbing which leads to part of the workpiece being worn away. While finishing a material often means polishing it to gain a smooth, reflective surface, the process can also involve roughening as in satin, matte or beaded finishes.
Abrasives are extremely commonplace and are used very extensively in a wide variety of industrial, domestic, and technological applications. This gives rise to a large variation in the physical and chemical composition of abrasives as well as the shape of the abrasive. Common uses for abrasives include grinding, polishing, buffing, honing, cutting, drilling, sharpening, lapping, and sanding (see abrasive machining). (For simplicity, "mineral" in this article will be used loosely to refer to both minerals and mineral-like substances whether man-made or not.)
Files are not abrasives. They remove material not by scratching or rubbing, but by the cutting action of sharp teeth which have been cut into the surface of the file, very much like those of a saw. However, diamond files are a form of coated abrasive (as they are metal rods coated with diamond powder).
Some naturally occurring abrasives are:
Abrasives - Applications Common applications for abrasives include the following:
uk online casino games
Adhesives
An adhesive is any substance applied to the surfaces of materials that binds them together and resists separation.[1] The term "adhesive" may be used interchangeably with glue, cement, mucilage, or paste.[2] Adjectives may be used in conjunction with the word “adhesive” to describe properties based on the substance's physical or chemical form, the type of materials joined, or conditions under which it is applied.
Adhesive Properties
Selecting a structural strength adhesive for a specific application requires performance criteria of several characteristics. First are bond-making properties that determine ease of use and in-place manufacturing cost:
Then there are the following cured bond properties:
Physical Properties
Application of adhesives
Adhesive for bonding structures or racks.
Adhesive for bonding the front, side and rear window glass.
Adhesive for bonding body roof structure.
Adhesive for bonding side panels of the structure.
Adhesive for bonding the floor.
Adhesive for bonding the cabins of the vehicles.
Adhesives for bonding different elements of the equipment.
Lubricants
A lubricant is a substance introduced to reduce friction between surfaces in mutual contact, which ultimately reduces the heat generated when the surfaces move. It may also have the function of transmitting forces, transporting foreign particles, or heating or cooling the surfaces. The property of reducing friction is known as lubricity.
In addition to industrial applications, lubricants are used for many other purposes. Other uses include cooking (oils and fats in use in frying pans, in baking to prevent food sticking), bio-medical applications on humans (e.g. lubricants for artificial joints), ultrasound examination, medical examinations, and the use of personal lubricant for sexual purposes.
Properties
A good lubricant generally possesses the following characteristics:
Applications
Lubricants perform the following key functions:
Ceramic
A ceramic is an inorganic, nonmetallic solid material comprising metal, nonmetal or metalloid atoms primarily held in ionic and covalent bonds.
What are ceramics?
Ceramics once referred purely to pottery and to articles made by firing materials extracted from Earth. Today, the term has a much broader definition. Ceramics are generally thought of as inorganic and nonmetallic solids with a range of useful properties, including very high hardness and strength, extremely high melting points, and good electrical and thermal insulation.
The best-known ceramics are pottery, glass, brick, porcelain, and cement. But the general definition of a ceramic—a nonmetallic and inorganic solid—is so broad that it covers a much wider range of materials. At one end of the scale, ceramics include simple materials such as graphite and diamond, made up from different crystalline arrangements of the element carbon. But at the other end of the scale, complex crystals of yttrium, barium, copper, and oxygen make up the advanced ceramics used in so-called high-temperature superconductors (materials with almost no electrical resistance). Most ceramics fall somewhere between these extremes. Many are metal oxides, crystalline compounds of a metal element and oxygen. Others are silicides, borides, carbides, and nitrides, respectively made from silicon, boron, carbon, and nitrogen. Some of the most advanced ceramic materials are combinations of ceramics and other materials known as ceramic matrix composites (CMCs).
Photo: Porcelain plates are very familiar examples of ceramics, but there are other, much more surprising uses of ceramics too.
Properties of ceramics
Ceramics are best known as brittle solids particularly suited for withstanding high temperatures but, in fact, the different materials used in ceramics can give them a wide range of properties. The classic properties of ceramics include durability, strength and brittleness, high electrical and thermal resistance, and an ability to withstand the damaging effects of acids, oxygen, and other chemicals because of their inertness (chemical unreactivity). But not all ceramics behave in this way. For example, graphite is a very soft ceramic and conducts electricity well, whereas diamond is a very good conductor of heat. Ceramics called ferrites are particularly good conductors of electricity and superconductors have almost no electrical resistance at all. Ceramic matrix composites, made by embedding fibers of a strengthening material in what is known as a ceramic matrix, are not at all brittle.
Photo: Silicon and carbon fuse to form silicon carbide power (left), which can be made into a hard and hard-wearing ceramic called silicon carbide that can survive high temperatures. It has many applications, from drills and cutting tools to components (middle, right) that can withstand high temperatures in gas-turbine engines that would melt ordinary metal parts. Ceramic components are also used in ordinary car engines for the same reason. Picture by Warren Gretz courtesy of US Department of Energy/National Renewable Energy Laboratory (NREL) (picture id 6307388).
The properties of a particular ceramic depend not just on the materials from which it is made but also on the way they are joined together—in other words, on its crystalline structure. Diamond is strong because all of its carbon atoms are bonded tightly to other carbon atoms. Graphite (such as that used in pencil "leads") shears because it is made up from different layers. Although the carbon atoms are tightly bonded within a given layer, the different layers are held together only by much weaker bonds. China clay (also called kaolin) behaves in a similar way to graphite, with its constituent aluminum, silicon, oxygen, and hydrogen atoms tightly bonded into flat sheets. But the weak bonds between those sheets are easily broken when water surrounds them and it is this that makes wet clay so easy to mold. When china clay is fired, heat removes the water, and the chemicals inside the clay rearrange themselves into crystals of aluminum silicate tightly bonded by silicate glass, which is overall very much stronger.
How ceramics are made
Firing is the process by which ceramics have traditionally been made; indeed, the word "ceramic" can be traced back to a Sanskrit word meaning "to burn." Simple ceramics such as bricks and certain types of glass are still made by processes that would be recognized by people who lived thousands of years ago. Just as in ancient times, today's pottery is made by digging clay from the ground, mixing it with water to make it flexible, shaping it on a wheel or in a mold, and then firing it in a kiln. Some of today's processes are more sophisticated than the techniques of past times. Machines have long been used in processes such as extrusion (forcing a material into shape by squeezing it like toothpaste through a shaped tool), jiggering (laying the material automatically into a rotating mold), or hot pressing (forcing a powdered form of the ceramic into a mold then simultaneously heating it and pressing it to fuse the material into shape)
Refractories
A refractory material is one that retains its strength at high temperatures. ASTM C71 defines refractories as "non-metallic materials having those chemical and physical properties that make them applicable for structures, or as components of systems, that are exposed to environments above 1,000 °F (811 K; 538 °C)"
Refractory materials are used in linings for furnaces, kilns, incinerators and reactors. They are also used to make crucibles and moulds for casting glass and metals and for surfacing flame deflector systems for rocket launch structures
Refractory materials
Refractory materials must be chemically and physically stable at high temperatures. Depending on the operating environment, they need to be resistant to thermal shock, be chemically inert, and/or have specific ranges of thermal conductivity and of the coefficient of thermal expansion.
The oxides of aluminium (alumina), silicon (silica) and magnesium (magnesia) are the most important materials used in the manufacturing of refractories. Another oxide usually found in refractories is the oxide of calcium (lime). Fire clays are also widely used in the manufacture of refractories.
Refractories must be chosen according to the conditions they will face. Some applications require special refractory materials. Zirconia is used when the material must withstand extremely high temperatures. Silicon carbide and carbon (graphite) are two other refractory materials used in some very severe temperature conditions, but they cannot be used in contact with oxygen, as they will oxidize and burn.
Binary compounds such as tungsten carbide or boron nitride can be very refractory. Hafnium carbide is the most refractory binary compound known, with a melting point of 3890 °C. The ternary compound tantalum hafnium carbide has one of the highest melting points of all known compounds (4215 °C).
applications
An abrasive is a material, often a mineral, that is used to shape or finish a workpiece through rubbing which leads to part of the workpiece being worn away. While finishing a material often means polishing it to gain a smooth, reflective surface, the process can also involve roughening as in satin, matte or beaded finishes.
Abrasives are extremely commonplace and are used very extensively in a wide variety of industrial, domestic, and technological applications. This gives rise to a large variation in the physical and chemical composition of abrasives as well as the shape of the abrasive. Common uses for abrasives include grinding, polishing, buffing, honing, cutting, drilling, sharpening, lapping, and sanding (see abrasive machining). (For simplicity, "mineral" in this article will be used loosely to refer to both minerals and mineral-like substances whether man-made or not.)
Files are not abrasives. They remove material not by scratching or rubbing, but by the cutting action of sharp teeth which have been cut into the surface of the file, very much like those of a saw. However, diamond files are a form of coated abrasive (as they are metal rods coated with diamond powder).
Some naturally occurring abrasives are:
- Calcite (calcium carbonate)
- Emery (impure corundum)
- Diamond dust (synthetic diamonds are used extensively)
- Novaculite
- Pumice
- Rouge
- Sand
- Corundum
- Garnet
- Sandstone
- Tripoli
- Powdered Feldspar
- Staurolite
- Borazon (cubic boron nitride or CBN)
- Ceramic
- Ceramic aluminium oxide
- Ceramic iron oxide
- Corundum (alumina or aluminium oxide)
- Dry ice
- Glass powder
- Steel abrasive
- Silicon carbide (carborundum)
- Zirconia alumina
- Boron carbide
- Slags
Abrasives - Applications Common applications for abrasives include the following:
- Buffing
- Honing
- Drilling
- Grinding
- Sanding
- Polishing
- Cutting
- Sharpening
uk online casino games
- It can be used as control cables in bicycle brakes, car parking brakes & chokes, etc
- Abrasives are used in flexible steel hoses like shower hoses, armored cables, etc
- It is used in collets for chucks
- Abrasives are used in diesel injection needles
- It is used in valve stems such as internal combustion engine valves
- It is used in axle shafts
- Abrasives are used in hypodermic needles in medical, dental & veterinary
- It is used in tungsten rod for contact points
- It is used in precious metal contact points
- It is used in tungsten wire electrodes for lamp bulbs
- Abrasives are used in trigger components for firearms
- It is sprag bar for sprag clutches in automotive transmissions, elevators, helicopter transmissions
- It is used in precious tubes in solar panels, soldering irons, etc.
- It is used in spark plug electrodes
- It is used in copper rod for soldering guns
- It is used in piston rings
- It is used in cutting expensive metals
- It is used in laminated magnetic cores like transformer cores
- It is used in Ferrite Rods for Magnets
- It is used in high speed steel (twist drills, etc.)
- It is used in sewing needles
- It is used in wafering in various materials
- It is used in capillary tubes metal and glass
- It is used in tubing for extendable telescopic aerials
- It is used in thin walled tubes in aircraft, helicopters
Adhesives
An adhesive is any substance applied to the surfaces of materials that binds them together and resists separation.[1] The term "adhesive" may be used interchangeably with glue, cement, mucilage, or paste.[2] Adjectives may be used in conjunction with the word “adhesive” to describe properties based on the substance's physical or chemical form, the type of materials joined, or conditions under which it is applied.
Adhesive Properties
Selecting a structural strength adhesive for a specific application requires performance criteria of several characteristics. First are bond-making properties that determine ease of use and in-place manufacturing cost:
- Degree of surface preparation necessary
- Time to handling strength
- Cure conditions of heat or room temperature, the degree of pressure, and the fixturing to maintain that pressure
- Viscosity for pumping and staying in place after application. Pseudoplastic and thixotropic qualities are desireable so that the adhesive thins during the shearing action of delivery and thickens in place without further shearing.
- Application with automated bulk systems or hand-held applicator to meet varying production requirements.
Then there are the following cured bond properties:
Physical Properties
- Adhesion to a variety of substrates allows bonding of dissimilar materials if necessary
- High cohesive strength is desirable
- Flexibility improves peel strength by flexing with peel stress
- High elastic modulus of substrate and adhesive resists stress at the bond line
- High damping capacity of the adhesive dissipates dynamic stresses of vibration, motion, & impact throughout the bond & peel stresses at the bond line
- Flexibility and damping resistance resists thermal expansion stresses when the coefficients of thermal expansion are different between adhesive and substrates
- Resists end-use or post-processing temperatures to maintain adhesive chemistry and the physical bond
- Withstands physical shock at a range of temperatures
- Maintains adhesive performance despite exposure to UV light, rain, salt water, and other weathering conditions
- Ability to withstand degradation from diesel fuel, solvents and other chemicals
Application of adhesives
Adhesive for bonding structures or racks.
Adhesive for bonding the front, side and rear window glass.
Adhesive for bonding body roof structure.
Adhesive for bonding side panels of the structure.
Adhesive for bonding the floor.
Adhesive for bonding the cabins of the vehicles.
Adhesives for bonding different elements of the equipment.
Lubricants
A lubricant is a substance introduced to reduce friction between surfaces in mutual contact, which ultimately reduces the heat generated when the surfaces move. It may also have the function of transmitting forces, transporting foreign particles, or heating or cooling the surfaces. The property of reducing friction is known as lubricity.
In addition to industrial applications, lubricants are used for many other purposes. Other uses include cooking (oils and fats in use in frying pans, in baking to prevent food sticking), bio-medical applications on humans (e.g. lubricants for artificial joints), ultrasound examination, medical examinations, and the use of personal lubricant for sexual purposes.
Properties
A good lubricant generally possesses the following characteristics:
- high boiling point and low freezing point (in order to stay liquid within a wide range of temperature)
- high viscosity index
- thermal stability
- hydraulic stability
- demulsibility
- corrosion prevention
- high resistance to oxidation.
Applications
Lubricants perform the following key functions:
- Keep moving parts apart
- Reduce friction
- Transfer heat
- Carry away contaminants & debris
- Transmit power
- Protect against wear
- Prevent corrosion
- Seal for gases
- Stop the risk of smoke and fire of objects
- Prevent rust.
Ceramic
A ceramic is an inorganic, nonmetallic solid material comprising metal, nonmetal or metalloid atoms primarily held in ionic and covalent bonds.
What are ceramics?
Ceramics once referred purely to pottery and to articles made by firing materials extracted from Earth. Today, the term has a much broader definition. Ceramics are generally thought of as inorganic and nonmetallic solids with a range of useful properties, including very high hardness and strength, extremely high melting points, and good electrical and thermal insulation.
The best-known ceramics are pottery, glass, brick, porcelain, and cement. But the general definition of a ceramic—a nonmetallic and inorganic solid—is so broad that it covers a much wider range of materials. At one end of the scale, ceramics include simple materials such as graphite and diamond, made up from different crystalline arrangements of the element carbon. But at the other end of the scale, complex crystals of yttrium, barium, copper, and oxygen make up the advanced ceramics used in so-called high-temperature superconductors (materials with almost no electrical resistance). Most ceramics fall somewhere between these extremes. Many are metal oxides, crystalline compounds of a metal element and oxygen. Others are silicides, borides, carbides, and nitrides, respectively made from silicon, boron, carbon, and nitrogen. Some of the most advanced ceramic materials are combinations of ceramics and other materials known as ceramic matrix composites (CMCs).
Photo: Porcelain plates are very familiar examples of ceramics, but there are other, much more surprising uses of ceramics too.
Properties of ceramics
Ceramics are best known as brittle solids particularly suited for withstanding high temperatures but, in fact, the different materials used in ceramics can give them a wide range of properties. The classic properties of ceramics include durability, strength and brittleness, high electrical and thermal resistance, and an ability to withstand the damaging effects of acids, oxygen, and other chemicals because of their inertness (chemical unreactivity). But not all ceramics behave in this way. For example, graphite is a very soft ceramic and conducts electricity well, whereas diamond is a very good conductor of heat. Ceramics called ferrites are particularly good conductors of electricity and superconductors have almost no electrical resistance at all. Ceramic matrix composites, made by embedding fibers of a strengthening material in what is known as a ceramic matrix, are not at all brittle.
Photo: Silicon and carbon fuse to form silicon carbide power (left), which can be made into a hard and hard-wearing ceramic called silicon carbide that can survive high temperatures. It has many applications, from drills and cutting tools to components (middle, right) that can withstand high temperatures in gas-turbine engines that would melt ordinary metal parts. Ceramic components are also used in ordinary car engines for the same reason. Picture by Warren Gretz courtesy of US Department of Energy/National Renewable Energy Laboratory (NREL) (picture id 6307388).
The properties of a particular ceramic depend not just on the materials from which it is made but also on the way they are joined together—in other words, on its crystalline structure. Diamond is strong because all of its carbon atoms are bonded tightly to other carbon atoms. Graphite (such as that used in pencil "leads") shears because it is made up from different layers. Although the carbon atoms are tightly bonded within a given layer, the different layers are held together only by much weaker bonds. China clay (also called kaolin) behaves in a similar way to graphite, with its constituent aluminum, silicon, oxygen, and hydrogen atoms tightly bonded into flat sheets. But the weak bonds between those sheets are easily broken when water surrounds them and it is this that makes wet clay so easy to mold. When china clay is fired, heat removes the water, and the chemicals inside the clay rearrange themselves into crystals of aluminum silicate tightly bonded by silicate glass, which is overall very much stronger.
How ceramics are made
Firing is the process by which ceramics have traditionally been made; indeed, the word "ceramic" can be traced back to a Sanskrit word meaning "to burn." Simple ceramics such as bricks and certain types of glass are still made by processes that would be recognized by people who lived thousands of years ago. Just as in ancient times, today's pottery is made by digging clay from the ground, mixing it with water to make it flexible, shaping it on a wheel or in a mold, and then firing it in a kiln. Some of today's processes are more sophisticated than the techniques of past times. Machines have long been used in processes such as extrusion (forcing a material into shape by squeezing it like toothpaste through a shaped tool), jiggering (laying the material automatically into a rotating mold), or hot pressing (forcing a powdered form of the ceramic into a mold then simultaneously heating it and pressing it to fuse the material into shape)
Refractories
A refractory material is one that retains its strength at high temperatures. ASTM C71 defines refractories as "non-metallic materials having those chemical and physical properties that make them applicable for structures, or as components of systems, that are exposed to environments above 1,000 °F (811 K; 538 °C)"
Refractory materials are used in linings for furnaces, kilns, incinerators and reactors. They are also used to make crucibles and moulds for casting glass and metals and for surfacing flame deflector systems for rocket launch structures
Refractory materials
Refractory materials must be chemically and physically stable at high temperatures. Depending on the operating environment, they need to be resistant to thermal shock, be chemically inert, and/or have specific ranges of thermal conductivity and of the coefficient of thermal expansion.
The oxides of aluminium (alumina), silicon (silica) and magnesium (magnesia) are the most important materials used in the manufacturing of refractories. Another oxide usually found in refractories is the oxide of calcium (lime). Fire clays are also widely used in the manufacture of refractories.
Refractories must be chosen according to the conditions they will face. Some applications require special refractory materials. Zirconia is used when the material must withstand extremely high temperatures. Silicon carbide and carbon (graphite) are two other refractory materials used in some very severe temperature conditions, but they cannot be used in contact with oxygen, as they will oxidize and burn.
Binary compounds such as tungsten carbide or boron nitride can be very refractory. Hafnium carbide is the most refractory binary compound known, with a melting point of 3890 °C. The ternary compound tantalum hafnium carbide has one of the highest melting points of all known compounds (4215 °C).
applications
- Burner Blocks
- Checker Wall Blocks
- Burner Pipe Covers
- Hearth Shapes
- Delta Sections
- Impact Pads
- Burner Ports
- Flare Tips
- Skimmer Blocks
- Troughs
- Pier Blocks
- Wall Panels
- Divider Walls
- Jambs, Lintel & Sills
- Well Blocks
- Flue Caps
- End Caps
- Roof Sections
- Safety Linings
- Runners
- Manway Plugs
- Air Grid Tiles
- Curb Blocks
- Peep Sites
- Nozzles
- Flue Walls
- Exhaust Ports
- Spouts
- Tap Blocks
- Sleeves
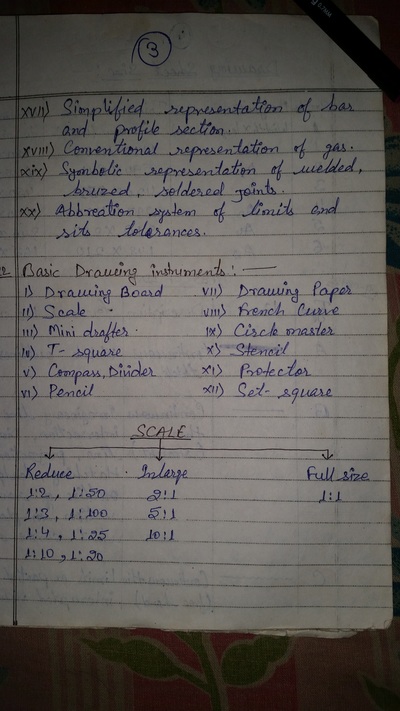
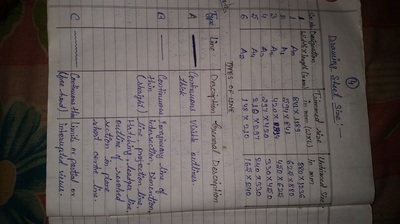
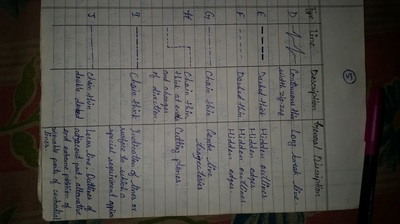
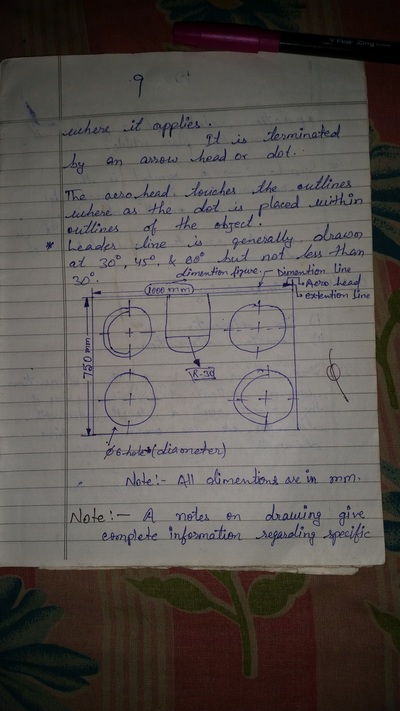
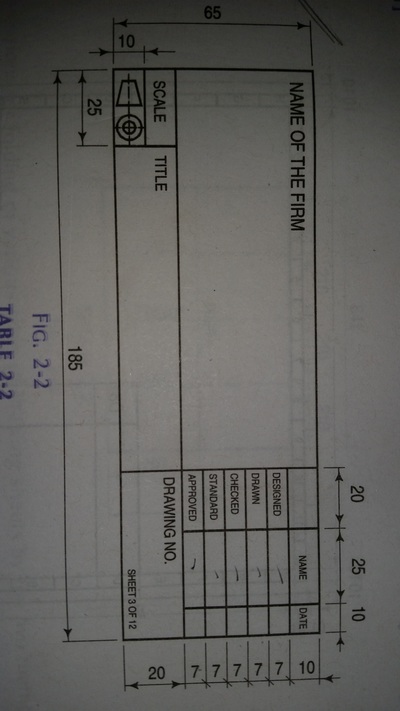
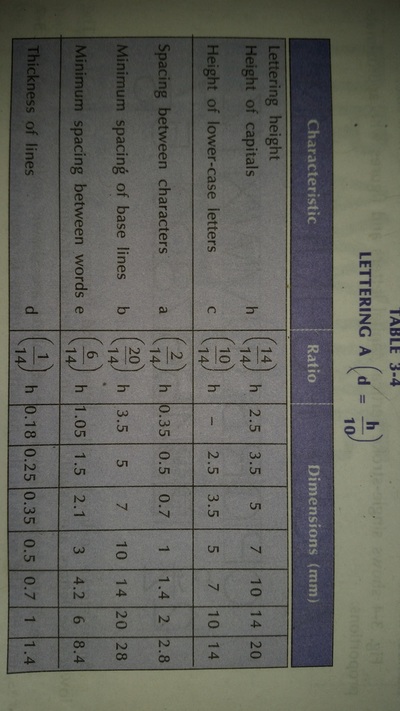
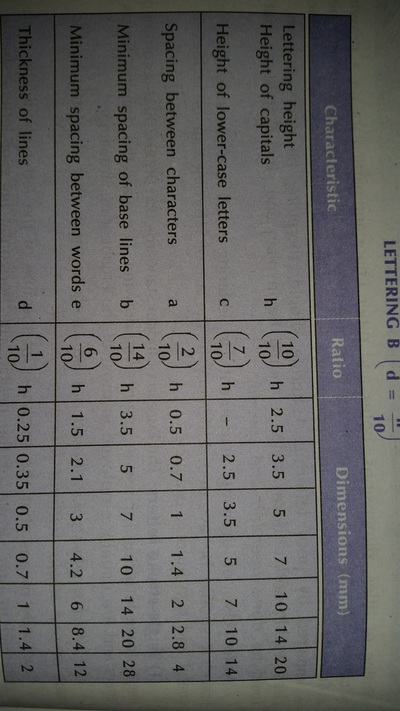
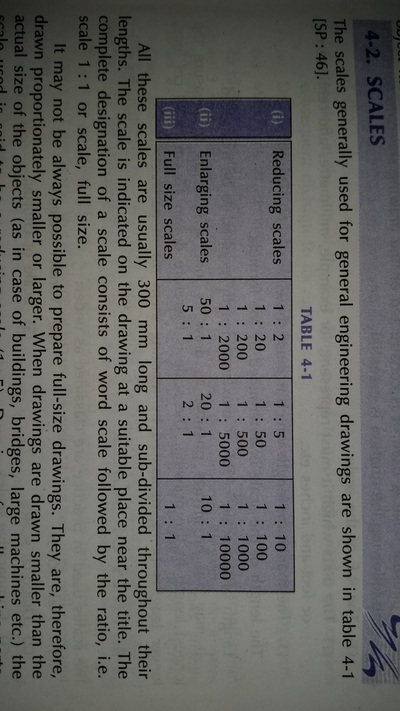
Engineering Drawing-1
Unit -1
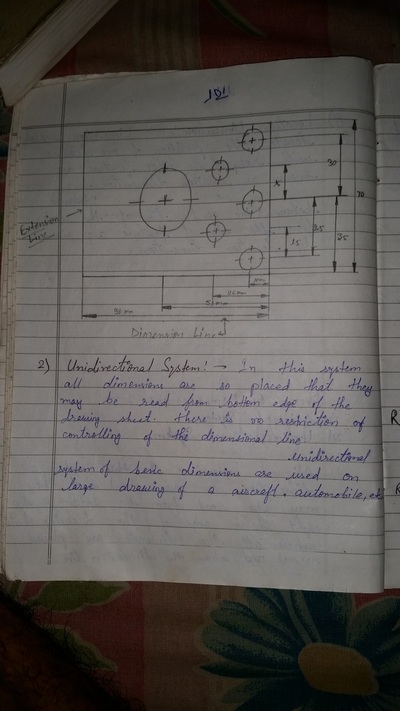
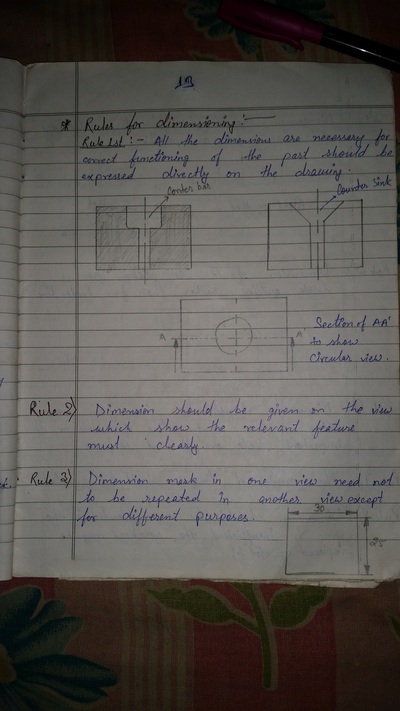
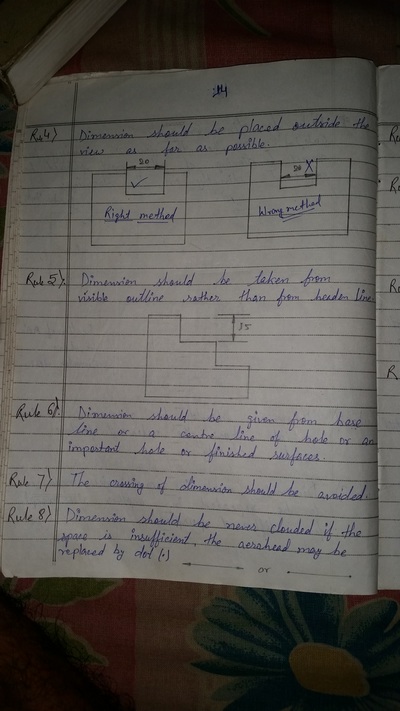
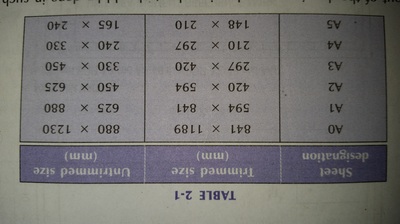
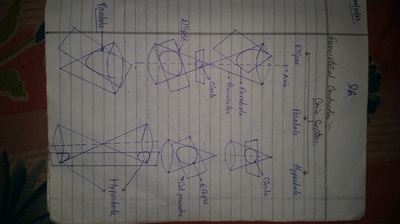
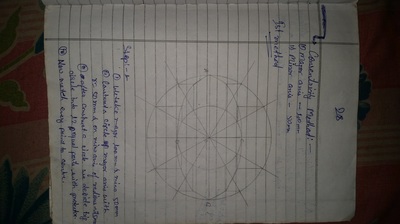
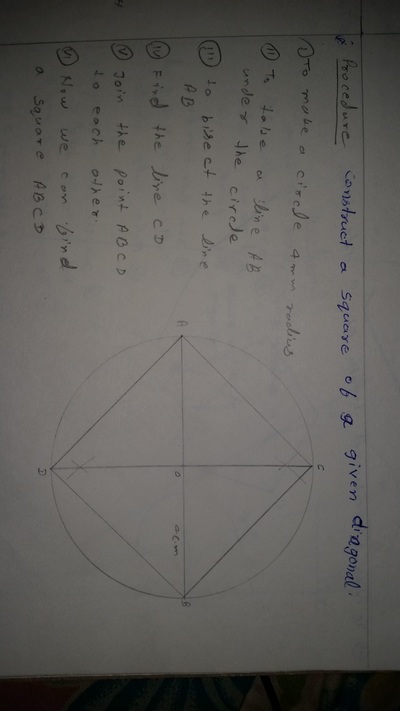
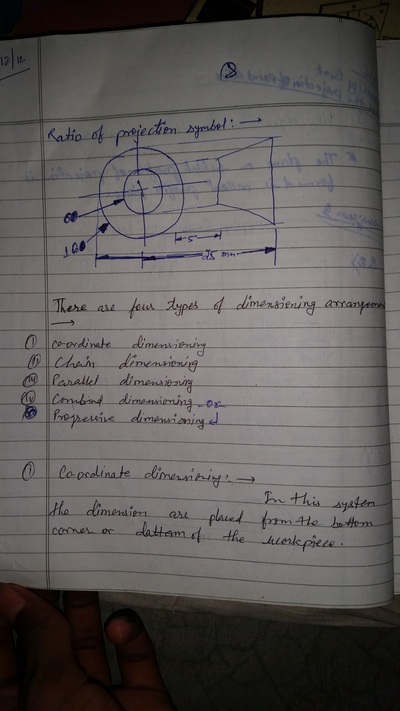
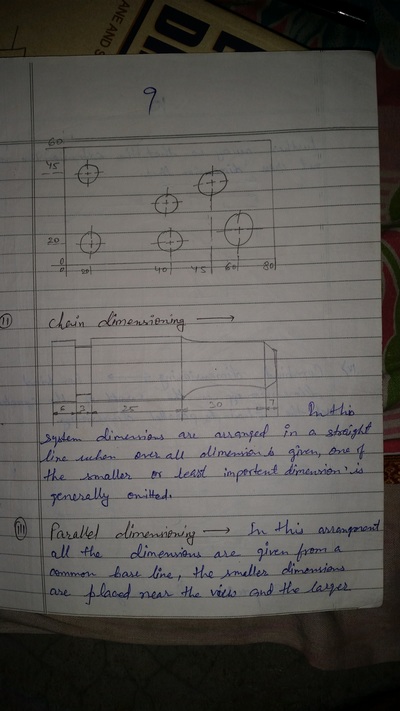
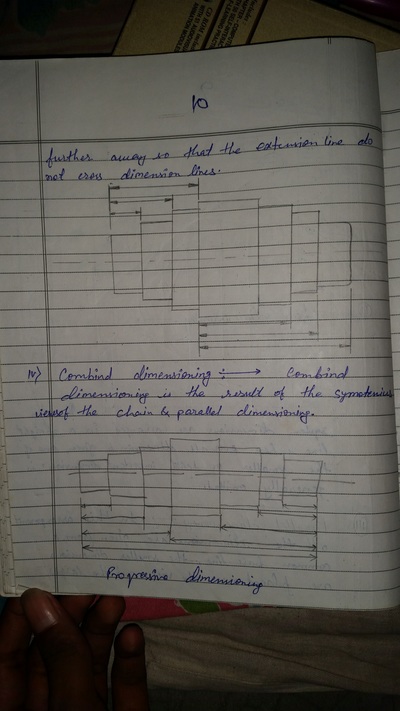
The importance of engineering Graphics, Drawing instruments, Drawing Standers, dimension, geometric construction
| geometric_construction.pdf | |
| File Size: | 486 kb |
| File Type: | |
Unit-2
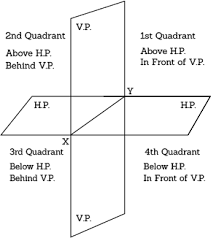
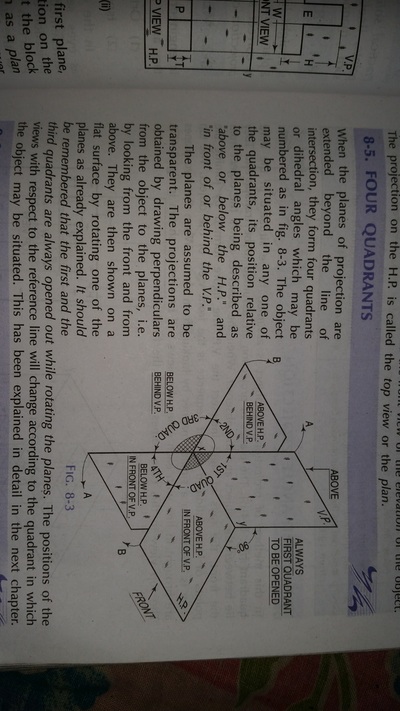
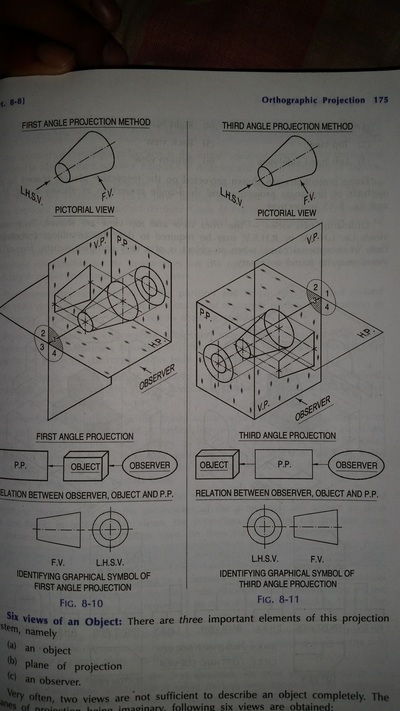
Principle of Orthographic Projection
| orthographic_projection.pdf | |
| File Size: | 693 kb |
| File Type: | |
Unit-3
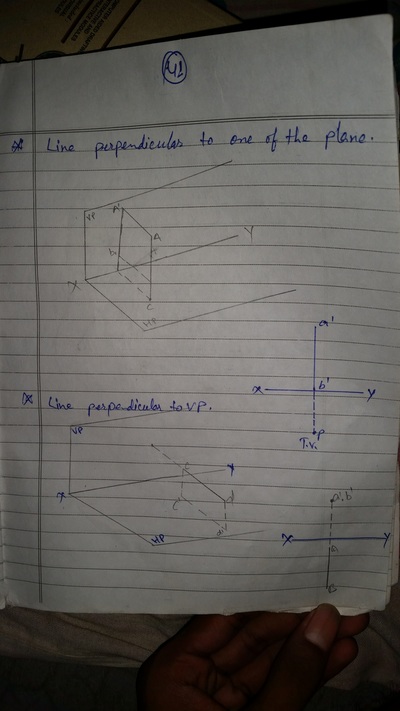
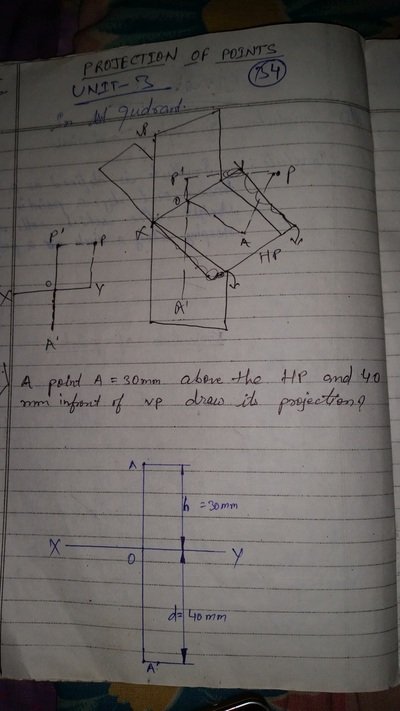
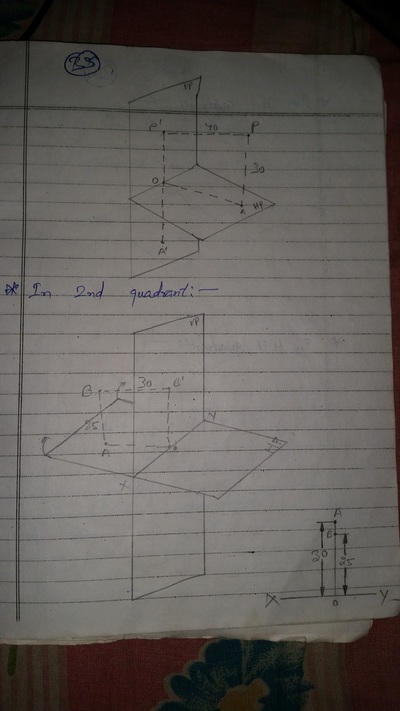
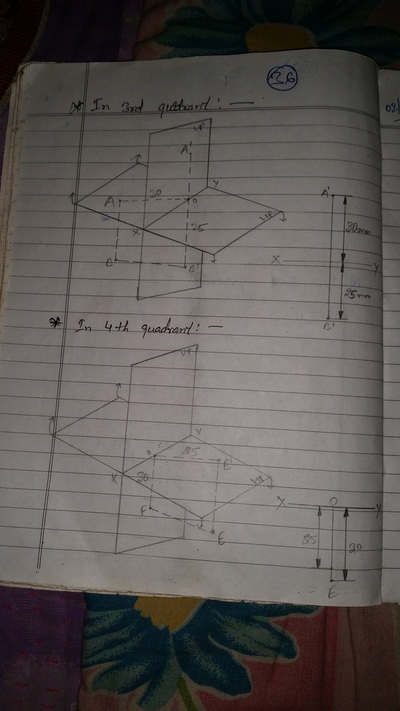
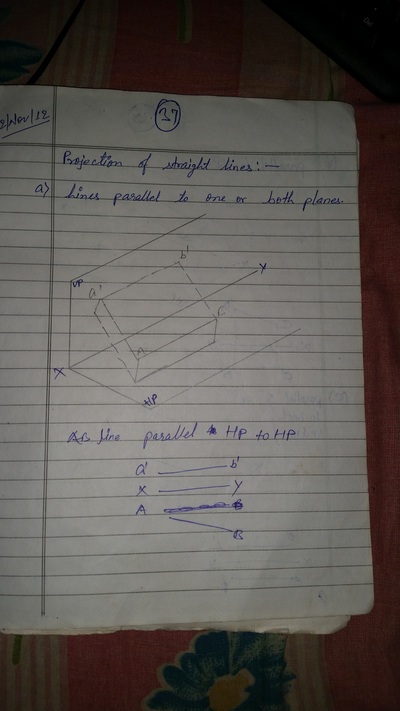
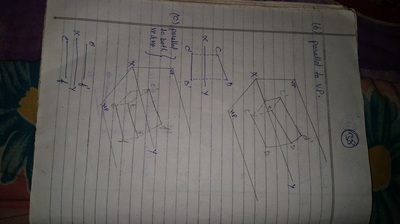
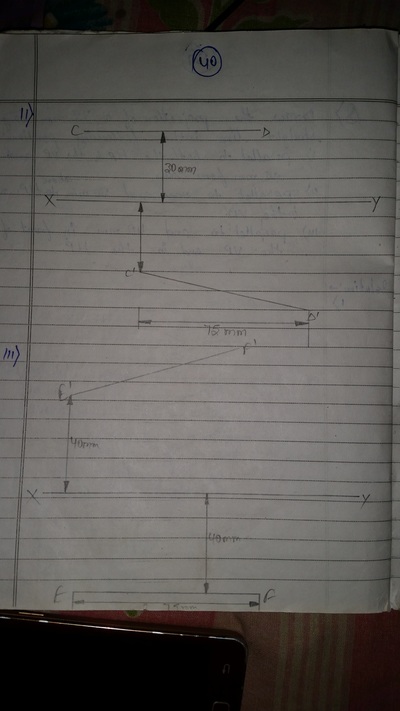
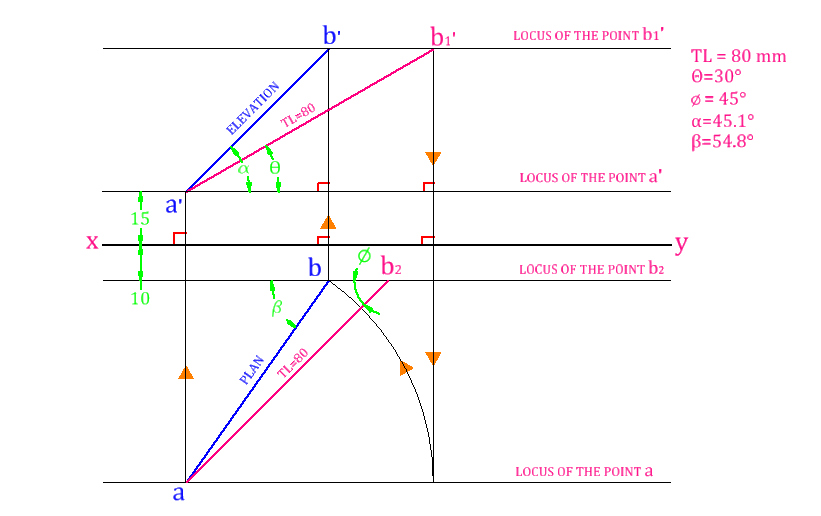
Projection of Points, Lines & Planes
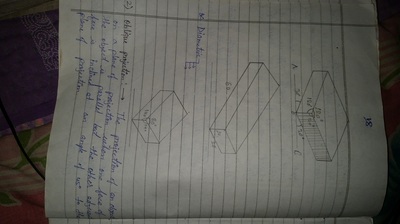
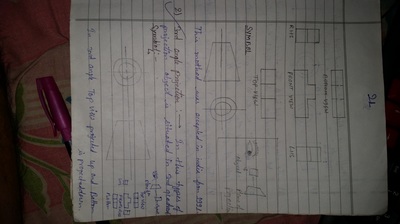
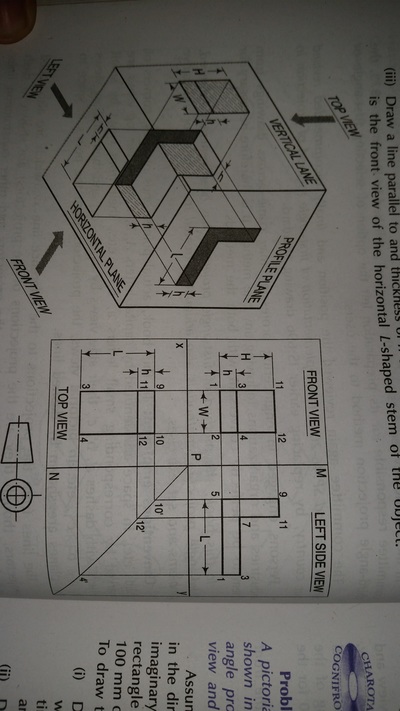
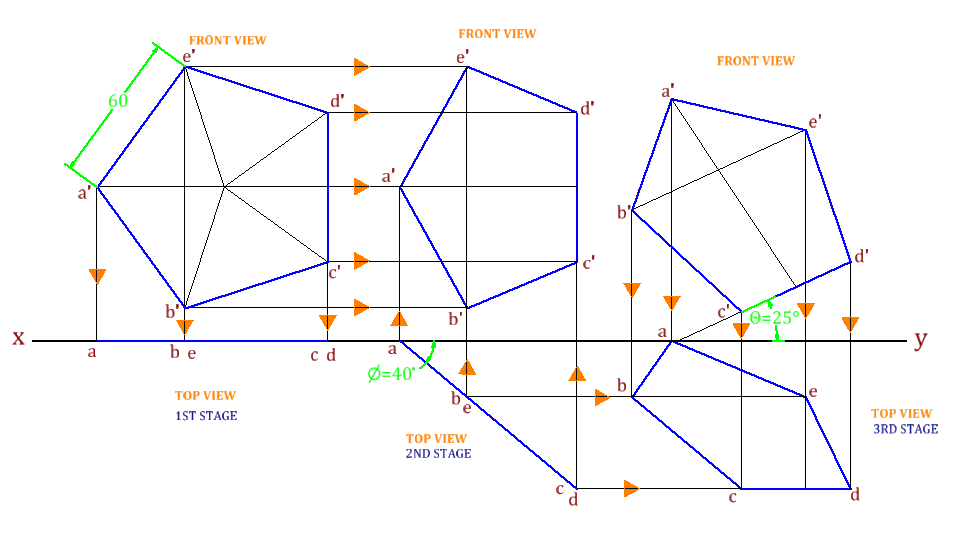
Unit-4
Projection of Solids
| unit-4_projection-of-solids.ppt | |
| File Size: | 814 kb |
| File Type: | ppt |
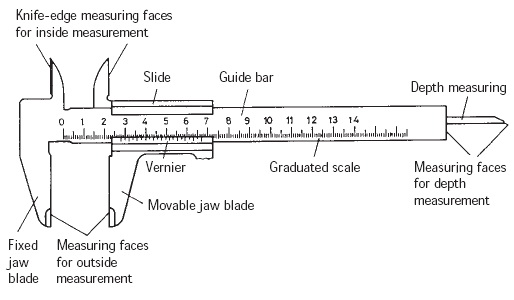
Physics Lab
Chemistry Lab
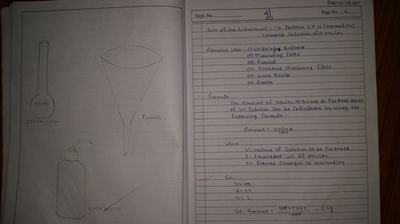
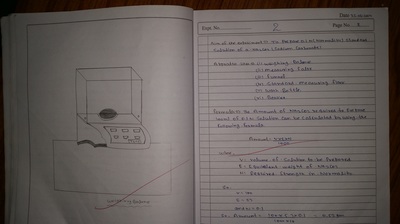
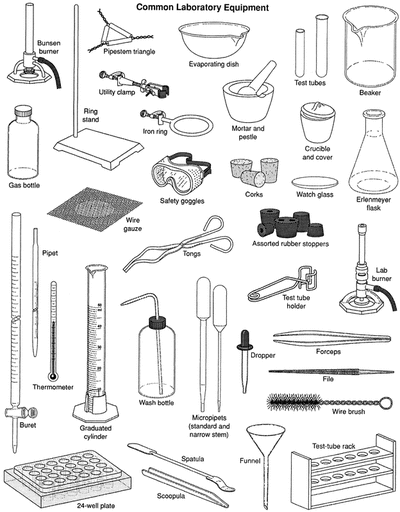
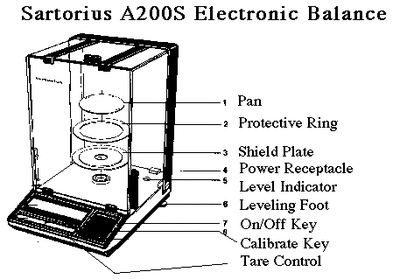
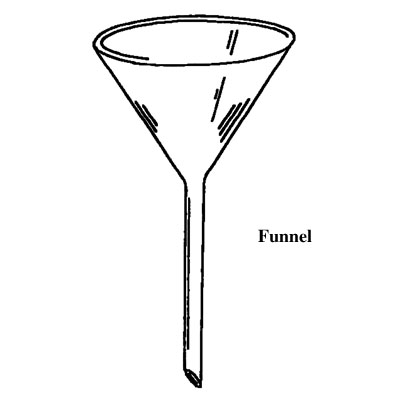
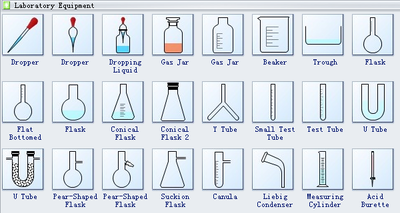
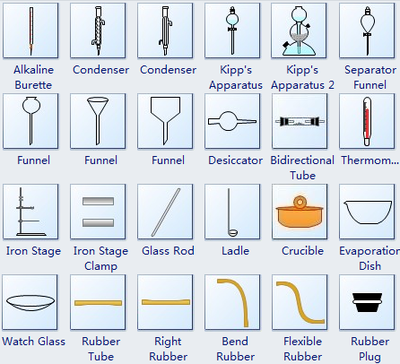
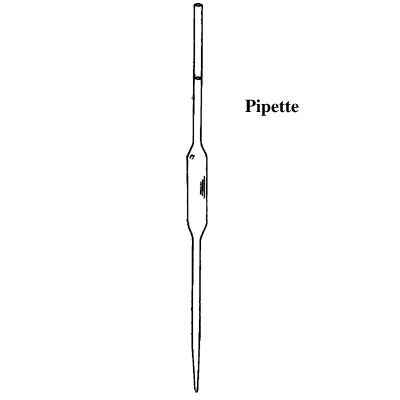
Apparatus used in chemistry lab
_