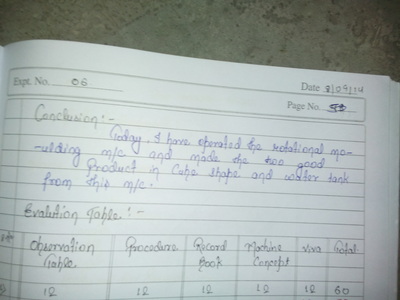
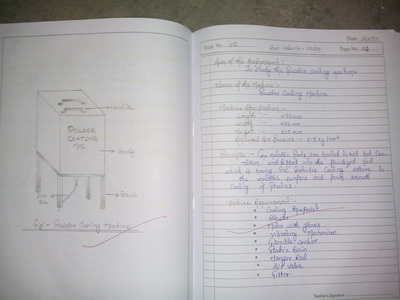
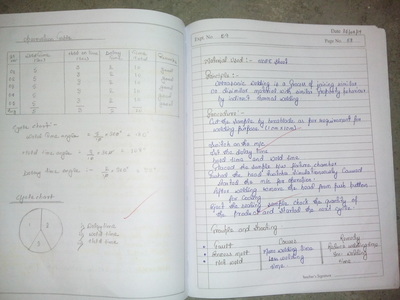
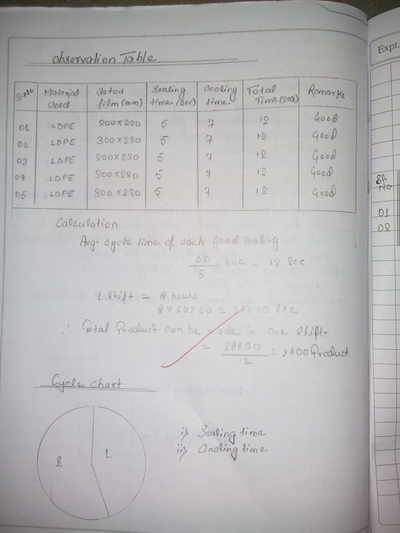
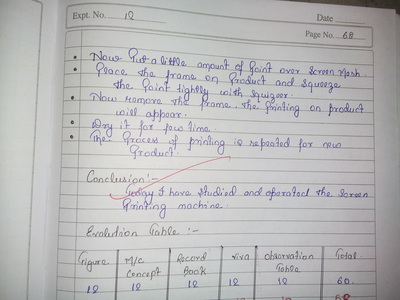
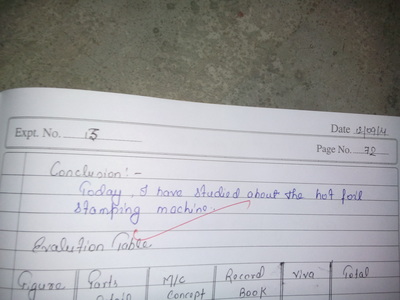
There is a feedback form at the bottom of this page. Please must fill it out.
इस पृष्ठ के नीचे एक फीडबैक फॉर्म है। कृपया इसे अवश्य भरें।
इस पृष्ठ के नीचे एक फीडबैक फॉर्म है। कृपया इसे अवश्य भरें।
Advanced plastics processing techniques
Unit-I (Specialized injection moulding process)
Advanced injection moulding process
All electric Injection moulding
Co-Injection moulding
| co-injection..ppt | |
| File Size: | 294 kb |
| File Type: | ppt |
2 colour Injection moulding
| |||||||
Gas assisted injection moulding
| |||||||
Water assisted injection moulding
Reaction injection moulding
| rim.pptx | |
| File Size: | 332 kb |
| File Type: | pptx |
Liquid injection moulding
Lost core injection moulding
Structural foam moulding
Thin wall injection moulding
Tie bar less injection moulding
Unit-II (Advanced blow moulding process)
|
| ||||||||||||
Unit-III (Advanced extrusion process)
|
| ||||||||||||||||||||||||||||||||||||
Maintenance of plastics processing & testing equipment
Unit-I
| intro_to_maintenance.ppt | |
| File Size: | 485 kb |
| File Type: | ppt |
| maintenance_definition .ppt | |
| File Size: | 193 kb |
| File Type: | ppt |
| maintenance_planning_and_scheduling--.ppt | |
| File Size: | 155 kb |
| File Type: | ppt |
Unit-II
| Alignment of machines .pdf | |
| File Size: | 1273 kb |
| File Type: | |
| levelling_introduction..ppt | |
| File Size: | 699 kb |
| File Type: | ppt |
Unit-III
| hydraulic_motors..ppt | |
| File Size: | 3167 kb |
| File Type: | ppt |
| basic_motor_maintenance..ppt | |
| File Size: | 1671 kb |
| File Type: | ppt |
Unit-IV
|
| ||||||||||||
|
| ||||||||||||
|
| ||||||||||||||||||||||||
Mould manufacturing techniques
Unit-I
Material for moulds
Alloying Elements
EFFECTS OF ALLOYING ELEMENTS
Effects of Alloying Elements in Steel
Alloying elements are added to effect changes in the properties of steels. The basis of this
section is to cover some of the different alloying elements added to the basic system of
iron and carbon, and what they do to change the properties or effectiveness of steel.
Carbon
As I've already stated, the presence of carbon in iron is necessary to make steel. Carbon is
essential to the formation of cementite (as well as other carbides), and to the formation of
pearlite, spheroidite, bainite, and iron-carbon martensite, with martensite being the
hardest of the micro-structures, and the structure sought after by knifemakers. The
hardness of steel (or more accurately, the hardenability) is increased by the addition of
more carbon, up to about 0.65 percent. Wear resistance can be increased in amounts up to
about 1.5 percent. Beyond this amount, increases of carbon reduce toughness and
increase brittleness. The steels of interest to knifemakers generally contain between 0.5
and 1.5 percent carbon. They are described as follows:
• Low Carbon: Under 0.4 percent
• Medium Carbon: 0.4 - 0.6 percent
• High Carbon: 0.7 - 1.5 percent
Carbon is the single most important alloying element in steel.
Manganese
Manganese slightly increases the strength of ferrite, and also increases the hardness
penetration of steel in the quench by decreasing the critical quenching speed. This also
makes the steel more stable in the quench. Steels with manganese can be quenched in oil
rather than water, and therefore are less susceptible to cracking because of a reduction in
the shock of quenching. Manganese is present in most commercially made steels.
Chromium
As with manganese, chromium has a tendency to increase hardness penetration. This
element has many interesting effects on steel. When 5 percent chromium or more is used
in conjunction with manganese, the critical quenching speed is reduced to the point that
the steel becomes air hardening. Chromium can also increase the toughness of steel, as
well as the wear resistance. Probably one of the most well known effects of chromium on
steel is the tendency to resist staining and corrosion. Steels with 14 percent or more
chromium are referred to as stainless steels. A more accurate term would be stain
resistant. Stainless tool steels will in fact darken and rust, just not as readily as the nonstainless
varieties. Steels with chromium also have higher critical temperatures in heat
treatment.
Silicon
Silicon is used as a deoxidizer in the manufacture of steel. It slightly increases the
strength of ferrite, and when used in conjunction with other alloys can help increase the
toughness and hardness penetration of steel.
Nickel
Nickel increases the strength of ferrite, therefore increasing the strength of the steel. It is
used in low alloy steels to increase toughness and hardenability. Nickel also tends to help
reduce distortion and cracking during the quenching phase of heat treatment.
Molybdenum
Molybdenum increases the hardness penetration of steel, slows the critical quenching
speed, and increases high temperature tensile strength.
Vanadium
Vanadium helps control grain growth during heat treatment. By inhibiting grain growth it
helps increase the toughness and strength of the steel.
Tungsten
Used in small amounts, tungsten combines with the free carbides in steel during heat
treatment, to produce high wear resistance with little or no loss of toughness. High
amounts combined with chromium gives steel a property known as red hardness. This
means that the steel will not lose its working hardness at high temperatures. An example
of this would be tools designed to cut hard materials at high speeds, where the friction
between the tool and the material would generate high temperatures.
Copper
The addition of copper in amounts of 0.2 to 0.5 percent primarily improves steels
resistance to atmospheric corrosion. It should be noted that with respect to knife steels,
copper has a detrimental effect to surface quality and to hot-working behavior due to
migration into the grain boundaries of the steel.
Niobium
In low carbon alloy steels Niobium lowers the transition temperature and aids in a fine
grain structure. Niobium retards tempering and can decrease the hardenability of steel
because it forms very stable carbides. This can mean a reduction in the amount of carbon
dissolved into the austenite during heat treating.
Boron
Boron can significantly increase the hardenability of steel without loss of ductility. Its
effectiveness is most noticeable at lower carbon levels. The addition of boron is usually
in very small amounts ranging from 0.0005 to 0.003 percent.
Titanium
This element, when used in conjunction with Boron, increases the effectiveness of the
Boron in the hardenability of steel.
Some examples of Non-Ferrous Metals we deal with are:
Non-Ferrous Metals
Non-ferrous metals are metals that do not contain iron. There are two groups of metals; ferrous and non-ferrous. Ferrous metals contain iron, for example carbon steel, stainless steel (both alloys; mixtures of metals) and wrought iron. Non-ferrous metals don't contain iron, for example aluminium, brass, copper (which can be remembered as ABC) and titanium. You can also get non-ferrous metals as alloys eg, brass is an alloy of copper and zinc.
Nonferrous metals are specified for structural applications requiring reduced weight, higher strength, nonmagnetic properties, higher melting points, or resistance to chemical and atmospheric corrosion. They are also specified for electrical and electronic applications.
Aluminum
Pure aluminum is a silvery-white metal with many desirable characteristics. It is light, nontoxic (as the metal), nonmagnetic and nonsparking. It is easily formed, machined, and cast. Pure aluminum is soft and lacks strength, but alloys with small amounts of copper, magnesium, silicon, manganese, and other elements have very useful properties. Aluminum is an abundant element in the earth's crust, but it is not found free in nature. The Bayer process is used to refine aluminum from bauxite, an aluminum ore. Because of aluminum's mechanical and physical properties, it is an extremely convenient and widely used metal.
Some Common Uses -
Building & Construction Industry:
Properties -
Copper Copper provides a diverse range of properties: good thermal and electrical conductivity, corrosion resistance, ease of forming, ease of joining, and color. However, copper and its alloys have relatively low strength-to-weight ratios and low strengths at elevated temperatures. Some copper alloys are also susceptible to stress-corrosion cracking unless they are stress relieved. Next to silver, copper is the next best electrical conductor. It is a yellowish red metal that polishes to a bright metallic luster. It is tough, ductile and malleable. Copper has a disagreeable taste and a peculiar smell. Copper is resistant to corrosion in most atmospheres including marine and industrial environments. It is corroded by oxidizing acids, halogens, sulphides and ammonia based solutions.
Beryllium Beryllium has one of the highest melting points of the light metals. The modulus of elasticity of beryllium is approximately 1/3 greater than that of steel. It has excellent thermal conductivity, is nonmagnetic and resists attack by concentrated nitric acid. It is highly permeable to X-rays, and neutrons are liberated when it is hit by alpha particles, as from radium or polonium (about 30 neutrons/million alpha particles). At standard temperature and pressures beryllium resists oxidation when exposed to air (although its ability to scratch glass is probably due to the formation of a thin layer of the oxide). Beryllium is a very light weight metal with a high modulus of elasticity (five times that of ultrahigh-strength steels), high specific heat, and high specific strength (strength to weight ratio).
Uses -
Beryllium is used as an alloying agent in the production of beryllium-copper because of its ability to absorb large amounts of heat. Beryllium-copper alloys are used in a wide variety of applications because of their electrical and thermal conductivity, high strength and hardness, nonmagnetic properties, along with good corrosion and fatigue resistance. These applications include the making of spot-welding electrodes, springs, non-sparking tools and electrical contacts.
Due to their stiffness, light weight, and dimensional stability over a wide temperature range, beryllium-copper alloys are also used in the defense and aerospace industries as light-weight structural materials in high-speed aircraft, missiles, space vehicles and communication satellites.
Thin sheets of beryllium foil are used with X-ray detection diagnostics to filter out visible light and allow only X-rays to be detected.
In the field of X-ray lithography beryllium is used for the reproduction of microscopic integrated circuits.
Because it has a low thermal neutron absorption cross section, the nuclear power industry uses this metal in nuclear reactors as a neutron reflector and moderator.
Beryllium is used in nuclear weapons for similar reasons. For example, the critical mass of a plutonium sphere is significantly reduced if the plutonium is surrounded by a beryllium shell.
It is, however, brittle, chemically reactive, expensive to refine and form, and its impact strength is low compared to values for most other metals.
Rubber
Rubbers are described as materials which show “elastic” properties. Such materials are generally long
chain molecules known as “polymers” and the combination of elastic and polymers has led to the
alternative name of “elastomers”. Rubbers and elastomers will be considered to be synonymous in this
work.
Products made from rubber have a flexible and stable 3–dimensional chemical structure and are able
to withstand under force large deformations. For example the material can be stretched repeatedly to
at least twice its original length and, upon immediate release of the stress, will return with force to
approximately its original length.
Rubber is used as a name for 3 categories:
Raw or base polymers These determine the main characteristics of the final product.
Semi-manufactured product
The addition to raw rubber of various chemicals, to impart desirable properties, is termed compounding. This semi-finished material is getting its rubber properties after vulcanization.
Final product After moulding the rubber compounds gets its elastic properties after a
vulcanization process.
EFFECTS OF ALLOYING ELEMENTS
Effects of Alloying Elements in Steel
Alloying elements are added to effect changes in the properties of steels. The basis of this
section is to cover some of the different alloying elements added to the basic system of
iron and carbon, and what they do to change the properties or effectiveness of steel.
Carbon
As I've already stated, the presence of carbon in iron is necessary to make steel. Carbon is
essential to the formation of cementite (as well as other carbides), and to the formation of
pearlite, spheroidite, bainite, and iron-carbon martensite, with martensite being the
hardest of the micro-structures, and the structure sought after by knifemakers. The
hardness of steel (or more accurately, the hardenability) is increased by the addition of
more carbon, up to about 0.65 percent. Wear resistance can be increased in amounts up to
about 1.5 percent. Beyond this amount, increases of carbon reduce toughness and
increase brittleness. The steels of interest to knifemakers generally contain between 0.5
and 1.5 percent carbon. They are described as follows:
• Low Carbon: Under 0.4 percent
• Medium Carbon: 0.4 - 0.6 percent
• High Carbon: 0.7 - 1.5 percent
Carbon is the single most important alloying element in steel.
Manganese
Manganese slightly increases the strength of ferrite, and also increases the hardness
penetration of steel in the quench by decreasing the critical quenching speed. This also
makes the steel more stable in the quench. Steels with manganese can be quenched in oil
rather than water, and therefore are less susceptible to cracking because of a reduction in
the shock of quenching. Manganese is present in most commercially made steels.
Chromium
As with manganese, chromium has a tendency to increase hardness penetration. This
element has many interesting effects on steel. When 5 percent chromium or more is used
in conjunction with manganese, the critical quenching speed is reduced to the point that
the steel becomes air hardening. Chromium can also increase the toughness of steel, as
well as the wear resistance. Probably one of the most well known effects of chromium on
steel is the tendency to resist staining and corrosion. Steels with 14 percent or more
chromium are referred to as stainless steels. A more accurate term would be stain
resistant. Stainless tool steels will in fact darken and rust, just not as readily as the nonstainless
varieties. Steels with chromium also have higher critical temperatures in heat
treatment.
Silicon
Silicon is used as a deoxidizer in the manufacture of steel. It slightly increases the
strength of ferrite, and when used in conjunction with other alloys can help increase the
toughness and hardness penetration of steel.
Nickel
Nickel increases the strength of ferrite, therefore increasing the strength of the steel. It is
used in low alloy steels to increase toughness and hardenability. Nickel also tends to help
reduce distortion and cracking during the quenching phase of heat treatment.
Molybdenum
Molybdenum increases the hardness penetration of steel, slows the critical quenching
speed, and increases high temperature tensile strength.
Vanadium
Vanadium helps control grain growth during heat treatment. By inhibiting grain growth it
helps increase the toughness and strength of the steel.
Tungsten
Used in small amounts, tungsten combines with the free carbides in steel during heat
treatment, to produce high wear resistance with little or no loss of toughness. High
amounts combined with chromium gives steel a property known as red hardness. This
means that the steel will not lose its working hardness at high temperatures. An example
of this would be tools designed to cut hard materials at high speeds, where the friction
between the tool and the material would generate high temperatures.
Copper
The addition of copper in amounts of 0.2 to 0.5 percent primarily improves steels
resistance to atmospheric corrosion. It should be noted that with respect to knife steels,
copper has a detrimental effect to surface quality and to hot-working behavior due to
migration into the grain boundaries of the steel.
Niobium
In low carbon alloy steels Niobium lowers the transition temperature and aids in a fine
grain structure. Niobium retards tempering and can decrease the hardenability of steel
because it forms very stable carbides. This can mean a reduction in the amount of carbon
dissolved into the austenite during heat treating.
Boron
Boron can significantly increase the hardenability of steel without loss of ductility. Its
effectiveness is most noticeable at lower carbon levels. The addition of boron is usually
in very small amounts ranging from 0.0005 to 0.003 percent.
Titanium
This element, when used in conjunction with Boron, increases the effectiveness of the
Boron in the hardenability of steel.
Some examples of Non-Ferrous Metals we deal with are:
- Aluminium & Aluminium Alloys.
- Copper.
- Brass.
- Lead.
- Zinc.
- Stainless Steel.
- Electrical Cable.
Non-Ferrous Metals
Non-ferrous metals are metals that do not contain iron. There are two groups of metals; ferrous and non-ferrous. Ferrous metals contain iron, for example carbon steel, stainless steel (both alloys; mixtures of metals) and wrought iron. Non-ferrous metals don't contain iron, for example aluminium, brass, copper (which can be remembered as ABC) and titanium. You can also get non-ferrous metals as alloys eg, brass is an alloy of copper and zinc.
Nonferrous metals are specified for structural applications requiring reduced weight, higher strength, nonmagnetic properties, higher melting points, or resistance to chemical and atmospheric corrosion. They are also specified for electrical and electronic applications.
Aluminum
Pure aluminum is a silvery-white metal with many desirable characteristics. It is light, nontoxic (as the metal), nonmagnetic and nonsparking. It is easily formed, machined, and cast. Pure aluminum is soft and lacks strength, but alloys with small amounts of copper, magnesium, silicon, manganese, and other elements have very useful properties. Aluminum is an abundant element in the earth's crust, but it is not found free in nature. The Bayer process is used to refine aluminum from bauxite, an aluminum ore. Because of aluminum's mechanical and physical properties, it is an extremely convenient and widely used metal.
Some Common Uses -
Building & Construction Industry:
- door and window frames
- wall cladding, roofing, awnings
- high tension power lines, wires, cables, busbars
- components for television, radios, refrigerators and air-conditioners
- beverage cans, bottle tops
- foil wrap, foil semi-rigid containers
- kettles and saucepans
- propellers
- airplane and vehicle body sheet
- gearboxes, motor parts
- tennis racquets, softball bats
- indoor and outdoor furniture
Properties -
- very lightweight (about 1/3 the mass of an equivalent volume of steel or copper) but with alloying can become very strong.
- excellent thermal conductor
- excellent electrical conductor (on a weight-for-mass basis, aluminium will conduct more than twice as much electricity as copper)
- highly reflective to radiant energy in the electromagnetic spectrum
- highly corrosion resistant in air and water (including sea water)
- highly workable and can be formed into almost any structural shape
- non-magnetic
- non-toxic
Copper Copper provides a diverse range of properties: good thermal and electrical conductivity, corrosion resistance, ease of forming, ease of joining, and color. However, copper and its alloys have relatively low strength-to-weight ratios and low strengths at elevated temperatures. Some copper alloys are also susceptible to stress-corrosion cracking unless they are stress relieved. Next to silver, copper is the next best electrical conductor. It is a yellowish red metal that polishes to a bright metallic luster. It is tough, ductile and malleable. Copper has a disagreeable taste and a peculiar smell. Copper is resistant to corrosion in most atmospheres including marine and industrial environments. It is corroded by oxidizing acids, halogens, sulphides and ammonia based solutions.
Beryllium Beryllium has one of the highest melting points of the light metals. The modulus of elasticity of beryllium is approximately 1/3 greater than that of steel. It has excellent thermal conductivity, is nonmagnetic and resists attack by concentrated nitric acid. It is highly permeable to X-rays, and neutrons are liberated when it is hit by alpha particles, as from radium or polonium (about 30 neutrons/million alpha particles). At standard temperature and pressures beryllium resists oxidation when exposed to air (although its ability to scratch glass is probably due to the formation of a thin layer of the oxide). Beryllium is a very light weight metal with a high modulus of elasticity (five times that of ultrahigh-strength steels), high specific heat, and high specific strength (strength to weight ratio).
Uses -
Beryllium is used as an alloying agent in the production of beryllium-copper because of its ability to absorb large amounts of heat. Beryllium-copper alloys are used in a wide variety of applications because of their electrical and thermal conductivity, high strength and hardness, nonmagnetic properties, along with good corrosion and fatigue resistance. These applications include the making of spot-welding electrodes, springs, non-sparking tools and electrical contacts.
Due to their stiffness, light weight, and dimensional stability over a wide temperature range, beryllium-copper alloys are also used in the defense and aerospace industries as light-weight structural materials in high-speed aircraft, missiles, space vehicles and communication satellites.
Thin sheets of beryllium foil are used with X-ray detection diagnostics to filter out visible light and allow only X-rays to be detected.
In the field of X-ray lithography beryllium is used for the reproduction of microscopic integrated circuits.
Because it has a low thermal neutron absorption cross section, the nuclear power industry uses this metal in nuclear reactors as a neutron reflector and moderator.
Beryllium is used in nuclear weapons for similar reasons. For example, the critical mass of a plutonium sphere is significantly reduced if the plutonium is surrounded by a beryllium shell.
It is, however, brittle, chemically reactive, expensive to refine and form, and its impact strength is low compared to values for most other metals.
Rubber
Rubbers are described as materials which show “elastic” properties. Such materials are generally long
chain molecules known as “polymers” and the combination of elastic and polymers has led to the
alternative name of “elastomers”. Rubbers and elastomers will be considered to be synonymous in this
work.
Products made from rubber have a flexible and stable 3–dimensional chemical structure and are able
to withstand under force large deformations. For example the material can be stretched repeatedly to
at least twice its original length and, upon immediate release of the stress, will return with force to
approximately its original length.
Rubber is used as a name for 3 categories:
Raw or base polymers These determine the main characteristics of the final product.
Semi-manufactured product
The addition to raw rubber of various chemicals, to impart desirable properties, is termed compounding. This semi-finished material is getting its rubber properties after vulcanization.
Final product After moulding the rubber compounds gets its elastic properties after a
vulcanization process.
Unit-II (Mould making techniques)
(I) Convectional
|
|
| ||||||||||||||||||||
(II) Special machine tools
| jig_boring_machine.ppt | |
| File Size: | 262 kb |
| File Type: | ppt |
| pantograph_instructions.pdf | |
| File Size: | 507 kb |
| File Type: | |
| electrical_discharge_machine..pdf | |
| File Size: | 35 kb |
| File Type: | |
| edm.pdf | |
| File Size: | 458 kb |
| File Type: | |
cnc machines
| product_manufacturing_cnc.ppt | |
| File Size: | 4700 kb |
| File Type: | ppt |
Unit-III (Mould polishing techniques)
Unit-IV (Mould maintenance)
| mould_maintenance..ppt | |
| File Size: | 260 kb |
| File Type: | ppt |
Plastics testing lab
Indian Standards
|
| ||||||||||||||||||||||||||||||